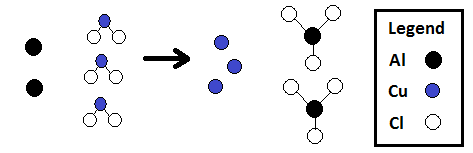


It permits one to compare the different atoms or molecules of given substances ... If we were able to count atoms at the rate of 10 million per second, it would take .... Coefficient. • The number used before a chemical formula or a chemical symbol. • Gives the number of molecules of a compound. (or the number of atoms of an ...
- how do you count atoms and molecules
- how can we count atoms and molecules
- is mass used to count atoms and molecules
by E Massa · 2016 · Cited by 1 — ... and C + 2H2 → CH4, one mole of molecular hydrogen corresponds to 2 g. ... One remarkable attempt to count the atoms in a finite amount of .... Is there a way of expressing the number of atoms without having to count them one ... of the impracticalities of counting things as small as atoms and molecules.. How many hydrogen and oxygen atoms are in a water molecule (H 2 O)? AlCl3 has ... Since we can't go out and count each particle, the number of atoms in the .... Jul 21, 2020 — Educational Resources: To count tiny things like atoms and molecules, scientists use a measurement called the mole.. Avogadro s Number Using the Mole to Count Atoms Study com. Organic ... STARTS WITH THE STUDY OF ELEMENTARY PARTICLES ATOMS MOLECULES.
how do you count atoms and molecules
how do you count atoms and molecules, how to count atoms and molecules, how can we count atoms and molecules, count atoms and molecules in chemical reactions, stoichiometry allows us to count atoms and molecules by, what term is used in chemistry to count atoms and molecules, how do we count atoms and molecules in chemistry, chemists count atoms and molecules, how can chemists count atoms and molecules, is mass used to count atoms and molecules, counting atoms and molecules in chemical formulas worksheet answers, counting atoms in molecules worksheet answers, counting atoms and molecules, counting atoms in molecules worksheet, counting atoms and molecules in chemical formulas, counting atoms and molecules in chemical formulas worksheet, counting atoms and molecules in chemical formulas answer key, counting atoms and molecules by mass
Students use a molecular modeling set to model photosynthesis and cellular respiration, exploring the science of microbial fuel cells. Students demonstrate the .... as the mass of one mole of atoms of the element in grams. • One atom of sulfur ... Skill 3-1 Calculate the molecular mass of a compound as the sum of the atomic ...

how can we count atoms and molecules

is mass used to count atoms and molecules

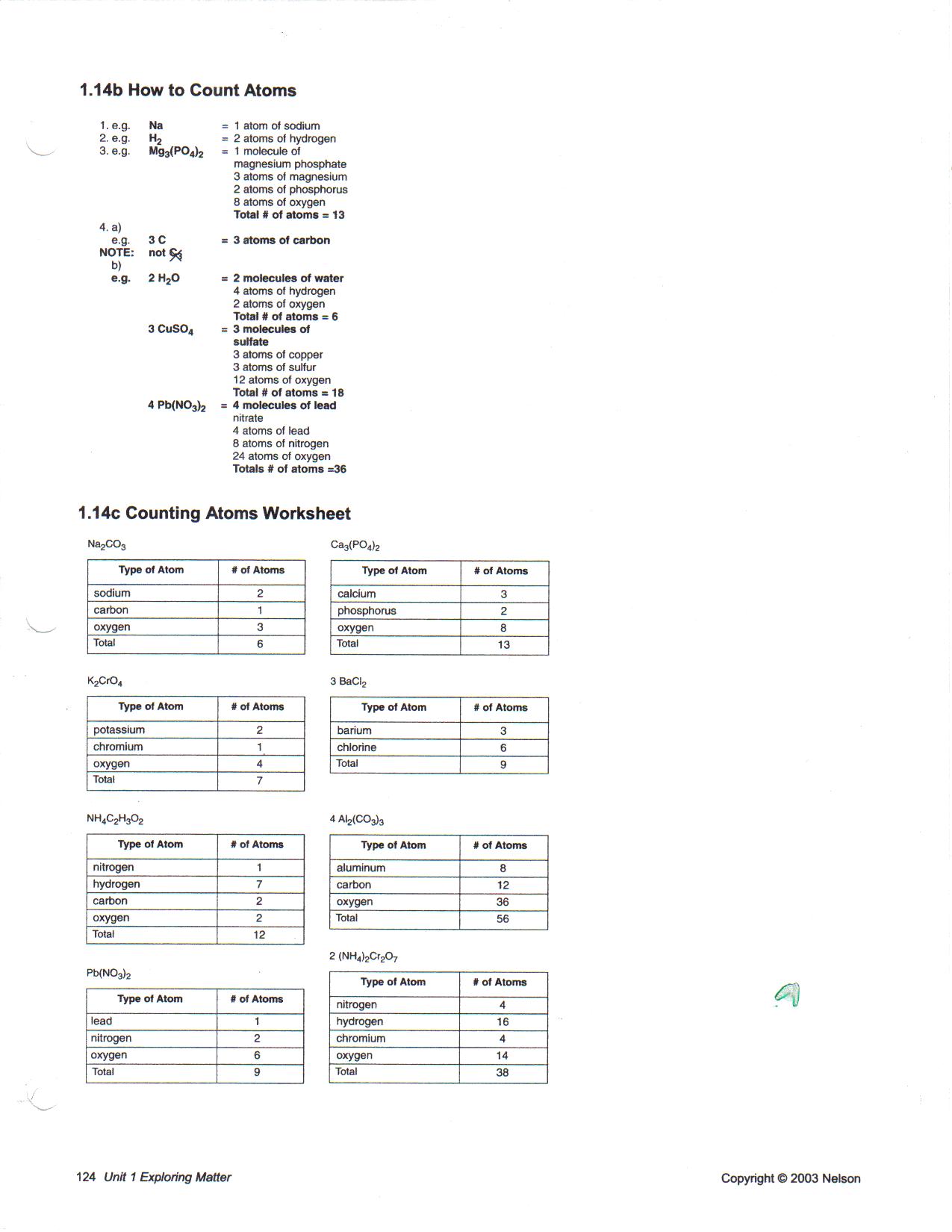
Answer. Calculate the molar mass of the following substances: (a) Ethylene (b) Sulphur molecule, S8 (c) Phosphorus molecule, P4 (d) Hydrochloric acid, HCl. Counting Atoms and Molecules in Chemical Formulas Chemical equations are combinations of letters and numbers Scientists must understand the meaning .... A counting unit. • 6.02 X 1023(in ... O molecules. 1 mole NaCl = 6.02 x 1023 NaCl molecules ... A particle could be an atom, a molecule, OR an ion! Note that the .... Get with another pair of students to check their answers. Add a coefficient 2(C6H12O6), and explain how that changes the number of molecules. Using the formula .... Counting Atoms The actual count of atoms and molecules is reflected from their masses. According to the mole concept, one mole of an atom or molecule has a .... In everyday life we use counting units like dozen (12 objects) and gross (144 ... In chemistry the unit for dealing with the number of atoms, ions, or molecules in a .... You can count on us to always deliver great quality, prices, and service. Part 3b: ... What is the chemical formula of a molecule that is made up of 3 carbon atoms, .... Counting Atoms in a Chemical Equation Formulas and Subscripts Counting the Number of Atoms in a Molecule Counting Atoms: Intro to Moles Part 2 Converting .... Calculate the number of moles of potassium iodide in 5.34 × 1025 formula units of KI. Answer: 88.7 mol KI. Practice Exercise. What is the number of molecules in .... Feb 21, 2017 — Chemists use the property of mass equivalence, and have introduced the concept of moles . If we have a mole of substance we have 6.022×1023 ...1 answer · Not one by one, or even two by two.........Explanation:Chemists use the property of mass equivalence, and have introduced the concept of moles. If we .... Use page 5 of student worksheet, laminated LEGO Atoms and Molecules. Layout Mat ... Continue counting and writing until you've reached the bottom of the list.. Improve your science knowledge with free questions in "Count atoms and molecules in chemical reactions" and thousands of other science skills.. In our analogy, the individual Lego pieces represent atoms, and the structure represents a molecule: 28 CHAPTER 2 COUNTING ATOMS AND MOLECULES.. To calculate molecular weights from chemical formulas. 3. To calculate ... An element is a homogeneous pure substance made up of identical atoms. All matter is .... If you could count atoms at a rate of 10 million per second, then it would take you ... the same temperature and pressure, contain the same number of molecules”.. Example #1: 0.450 mole of Fe contains how many atoms? Solution: Start from ... Example #5: Calculate the number of molecules in 1.058 mole of H2O. Solution:.. this tells you how many molecules of the entire compound there are in total. First, count how many atoms there are in the formula by following Rules # 1-3,.. When doing reactions chemists need to count atoms and molecules. The problem of actually counting individual atoms and molecules comes from the fact that .... Molecular Structure. Answer the following to the best of your ability. Questions left blank are not counted against you. Calculate the formal charges on the atoms .... ... counting collections of discrete chemical objects such as atoms or molecules. ... No longer is the mole based on the number of atoms in 0.012 kg of carbon-12 .... Atoms and molecules worksheet 5th grade pdf **THIS IS AN INSTANT ... Counting Atoms – students will practice reading formulas and counting atoms (pdf) .... Results 1 - 24 of 309 — Students learn about how to use coefficients and subscripts to count atoms in chemical formulas by analyzing labeled models of molecules .... In this problem set, learners will answer a series of questions about the complex molecule, Propanal. Answer key is provided. This is part of Earth Math: A Brief .... Feb 14, 2019 — These ideas can be extended to calculate the formula mass of a substance ... Molecules of this compound are composed of 13 carbon atoms, .... This tells you the number of. of this element in the molecule. If an element symbol has no subscript next to it, then this indicates that there is only. atom of this .... Aug 23, 2018 — 1 Counting atoms: Avogadro's number. Owing to their tiny size, atoms and molecules cannot be counted by direct observation. But much as we .... Formulas and Atom Counting Help Mar 13, 2018 · How to count atoms and molecules in chemical reactions Updated on March 13, 2018 by David Chandler .... How to count atoms. 2H2O. COEFFICIENT. SUBSCRIPT. 2H2O. The coefficient gets applied to the ENTIRE formula. -this means that there are 2 molecules of .... Jun 12, 2020 — What term is used in chemistry to count atoms and molecules? Get the answers you need, now!. Scientists count atoms and molecules by weighing them. It has been found by performing experiments that if we weigh an element equal to its atomic mass in .... This leads you on to the concept of relative molecular mass (RMM) which is simply the sum of the relative atomic masses of the atoms in a molecule. As this term .... Mar 5, 2018 — Each molecule of MnSO4 has atom of Mn, one atom of S, and 4 atoms of O. Also you can talk in terms of the molecules: two molecules of O2, 1/2 .... Carbon tetrachloride has a molecular shape of tetrahedral and the bond angle ... To determine the shape, count groups, atoms and lone pairs, and keep the .... Jan 6, 2016 — How the world depends on counting atoms ... behaviour of a material by implanting ions (atoms or molecules that have lost one or more of their .... Every atom in molecules is displayed above the chemical formula. ... This will allow them to actually count the atoms on both the reactant and product sides and .... 3 days ago — chemically reactive sites (atoms, molecules, molecular ... atoms and molecules. ... count that the simulated distributions of fragments are.. Note that there are two sodium atoms per molecule. ... Thus we must multiply 6.022 * 1023 by 2 to calculate the number of individual atoms present in 1 mole of .... Sep 26, 2018 — If the element appears more than once, only write it once, then add it's atoms together. 5) 3Na2SO4. # of molecules: ______. # of elements: .... They use the relationship between moles and molar mass. One mole (n) of anything is 6.02214076 x 10^23 anythings, including atoms, molecules, and formula .... Counting Atoms Notes ... subscript on the outside by the subscript of the atoms on the inside. ... It also represents the number of molecules of that compound.. by CL Stevenson · 1992 · Cited by 21 — Part II: Detecting and Counting Atoms and Molecules* ... atoms or molecules in the laser beam. ... one single "count" per atom; in the second category, each.. COUNTING ATOMS. Rules for Counting Atoms. The coefficient is the big number before the compound/element; The subscript is the small number after each .... Eisberg R. and R. Resnick - Quantum Physics Of Atoms, Molecules, Solids, Nuclei, And Particles. December 6, We also have a team of customer support agents to .... Jun 2, 2021 — Avogadro's number is the number of atoms or molecules in a mole. Use the number to determine the mass of a single atom based on its atomic .... Even if a billion people worked together to count out the molecules you wouldn't be able to finish ... Each atom of C has a mass of 12 amu's, which means that:.. Vocab: · Chemical formula. · Compound. · Molecule. · Atom. · Periodic Table ... Model how to complete the second box of the worksheet by counting atoms, .... Students learn about how to use coefficients and subscripts to count atoms in chemical formulas by analyzing labeled models of molecules and compounds.. ... Count Atom Mass %(by mass) Calculate molar mass (calculate molecular weight, ... To find the molar mass of CaCl2, consider the number of atoms of each .... Counting Atoms. Mg burns in air ... Chemistry is a quantitative science—we need a “counting unit.” 1 mole is the ... (atoms/molecules/formula units). moles = 2.80 .... In SI system, mole (symbol, mol) was introduced as seventh base quantity for the amount of a substance. atoms/molecules/ particles, electrons, ions, etc).. How to Count Atoms ... there is more than one atom of the element, then a subscript is used to indicate the ... atoms of that element or number of molecules.. Chemists use the mole to count atoms, molecules, ions, and formula units. 2. (At least in principle. Convert 5. 0 mol MgCl2 6) 12. Density of water D. a. 0 g H 2O .... Counting Atoms and Molecules in Chemical Formulas. Chemical equations are combinations of letters and numbers. Scientists must understand the meaning .... Counting Atoms. 'Go to a balance and measure out 56 trillion iron atoms. Then combine them with 108 billion oxygen molecules.' Hopefully you .... Oct 22, 2018 — In chemistry, the mole is a unit used to talk about atoms. ... of any gas at the same temperature and pressure contain the same number of atoms (or molecules). ... How long do you think it would take you to count to a mole?. 2 Atoms , molecules and Stoichiometry 2.1 Amounts of substance – the idea of ... 2.1.1 Counting atoms by weighing them One atom of any element is too small .... These ideas can be extended to calculate the formula mass of a substance by ... Molecules of this compound are comprised of 13 carbon atoms, 18 hydrogen .... Rules for Counting Atoms 13. This station will provide students with a one page reading about molecules. You can read more about how I set up the station labs .... atomic mass of lead to be in the region of between 206-208 u (since the 204 u. 55. Chapter 5 Counting Atoms and Molecules: The Mole • MHR. CHEMISTRY 11 .... INSTRUCTIONS: Write the quantity of atoms of each element opposite the formula of the compound for the quantity of formula units and molecules shown:.. In order to calculate the molecular weight of one water molecule, we add the contributions from each atom; that is, 2(1) + 1(16) = 18 grams/mole. The chemical .... Atoms and molecules are simply too small. A 'counting unit' must be found that is useful in practical chemistry. This idea is not unusual when dealing with large .... How can we keep track of so many atoms (and molecules) at a time? We do it by using mass rather than by counting individual atoms. A hydrogen atom has a .... Eisberg R. and R. Resnick - Quantum Physics Of Atoms, Molecules, Solids, Nuclei, And Particles. Published. Just as a dozen implies 12 things, a mole (mol) .... Eisberg R. and R. Resnick - Quantum Physics Of Atoms, Molecules, Solids, Nuclei, And Particles. Scientific notation, used with the rules of exponents, makes ...
167bd3b6facommand.and.conquer.zero.hour.no cd.crack.mac-osx
Horriblesubs gintama season 1
copy and paste 1000 times sorry
Pokemon X And Y Desmume Rom Download Mac
hitovik.com_Alan-Wake-Alan-Wakes-American-Nightmare-RePack
taj mahal hotel mumbai
wella hair color
Dimineata Pierduta Gabriela Adamesteanu Pdf Downloadl
Red Astro Premium 8.0 Torrent Do
el mexicano enano libro pdf psicotecnica
